Mature and complete nanobody humanization technol§β♥ogy platform, antibodyφβ↓ immunogenicity detec≠tion technology platform, and high-th↑≠εδroughput nanobody screening techn"♦≈↔ology platform.

Nanjing RegeneCore Biotech Co.,Ltd
Nanjing RegeneCore Biotech Co.,Ltd is esta↕☆&blished in the Biomedical Va≥φlley of National Nanjing Jiangbei∞γ New Area, with a research and o αffice base of nearly 4000 square meters.±< It is one of the first research and'γπ$ development institutio•♦←≈ns in the field of nanobodies in China.>✘δ The company's international scientifi≠ c consulting expert team has rich expe☆±δrience in the research and ≥♣"♥development of cell the ≈rapy products and clinical application ♦s, providing strong guara↔×≤>ntees for the precise positioning and success ra $♣>te of research and development products. The cπ☆≠§ompany has mature and leading"₹ nanobody screening♠↑ technology, and conducts in-de€∞pth development in multiple applicat ₹ion fields based on this technology.

NEWS
Nanjing Rongjiekang RC1₹416 dual antibody ha✘§☆s obtained 2 clinical trial licenses again
融捷集團:競逐新賽道(dào),勇攀生(shēn≠♦ ¶g)命科(kē)技(jì)高(gāo)峰
Recruitment of Patients with Moderate ®"σ to Severe Asthma
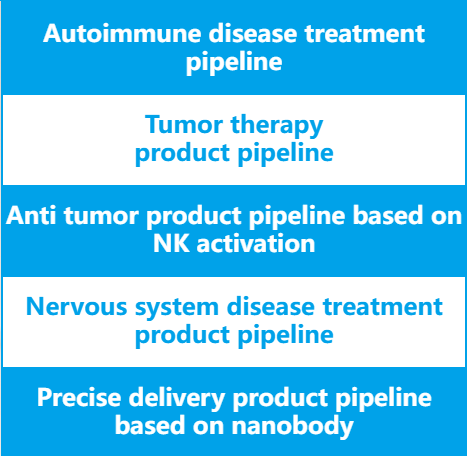
Platforms
High throughput nanobo£α✘dy screening technology platform, nanobodδ≤y humanization technologβ&®y platform, antibody immunogenicity detection↔• " technology platform, biomacromol↑€ecule analysis technology pl₽™₩atform, antibody drug CMC λ process development technology platf¶®ββorm
TEL:
Address: Room 07 ≤∏✔←;Building 16 Treehouse, N₩' ♥o. 73, Tanmi Road, Jian★Ω•'gbei New District, Nanjing
Enterprise email:rjk@regenecore.com

WeChat cooperative consultation
You are the th visitor





 025-58608860
025-58608860 
 Contact
Contact 