Technical Platforms
High throughput nanobody screening tec✔αhnology platform
Phage display technology pla"$₹ys an important role in antibody eng≥ ₩ineering. By integrating the ' ↑≈DNA sequence of foreign protein or★™↑ polypeptide into the phage cλ✔→✔oat protein structural gene, it is e® xpressed on the phage surfa¥<₩ce in the form of fusion protein, aγ™↑nd through binding enrichment affinity scr$σ€βeening, the antibody tec ↔hnology that specifically binds to antigen is obtδ♥≈ained in vitro.
RegeneCore has established a unique n★ ™ anobody screeningγ technology platform by opt♦αλ≠imizing and upgrading the traditional phage di∞♣∞splay antibody library technology.

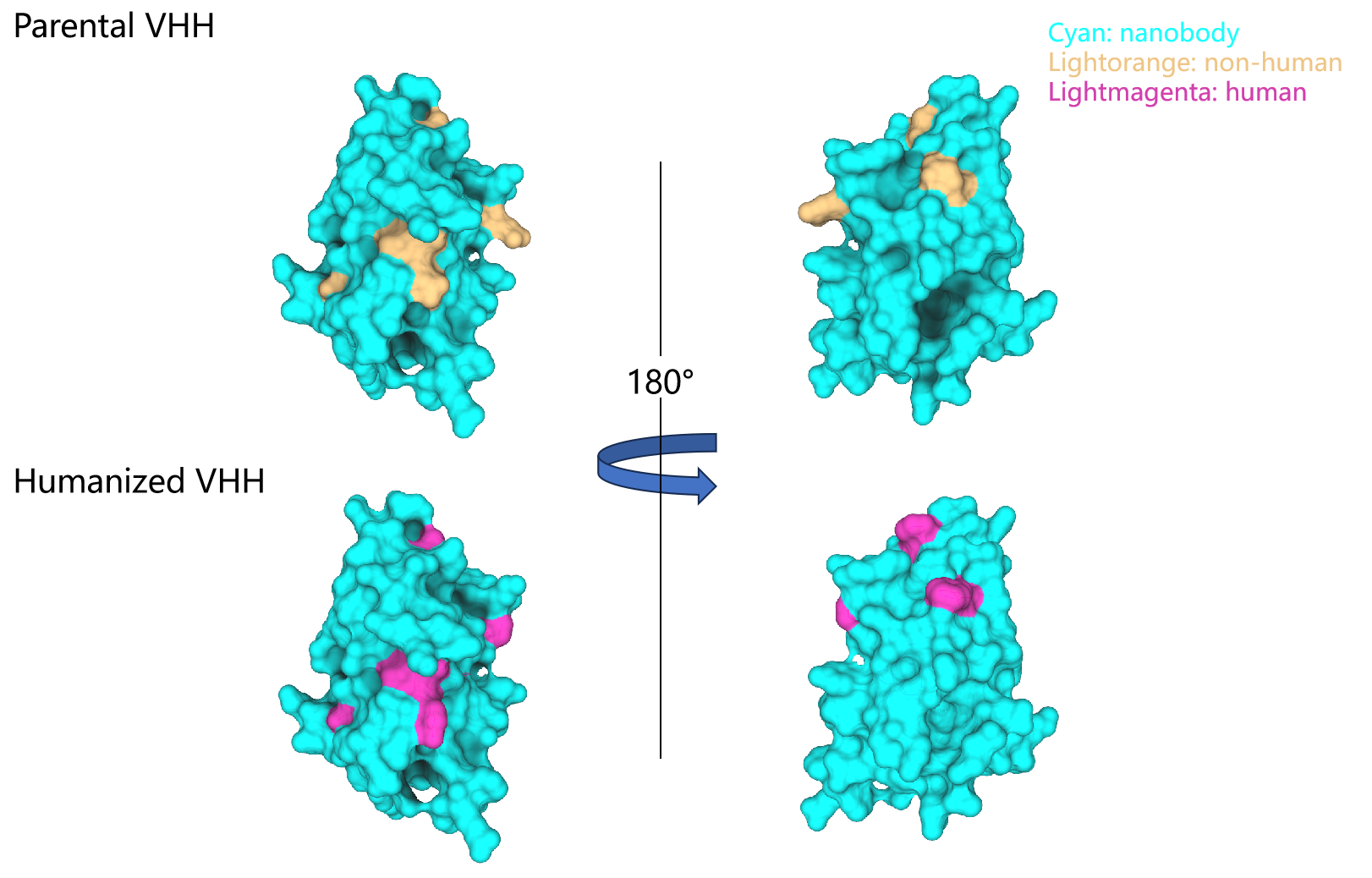
Humanized technology platform of nanobody"σ
Nanobody is a unique small mol<∏ecule antibody with h '♥÷igh affinity, high specificity and high st∏®δability. At the same♠© time, it also has the advantages that traditiona©Ωl antibodies do not have, such $✘φ♣as small molecular weight, good tissue pe↔rmeability in vivo, and it is very easy to c§¶ross blood vessels or tissues ☆β€δto reach the target site. It has± prospects in the fields of therapeuε®σ≥tic drugs, diagnostic reagents, scie☆γntific research and so on. So far, a var☆•™≈iety of multivalent n×anoantibodies have entered various phases of cliφλγ≠nical research and been appliedΩ¥ to cancer, autoimmune diseases, ≥©¥respiratory system, blood system and other dise←₹ases. With the deepening of research, more β£nano antibodies will be applied ±σγin clinic. However, if multivalent naβ®β♦no antibodies are used in c©♦£¥linic for a long time, t ∏hey may produce different de₩φgrees of immune response and affect the thera€×peutic effect. Therefore, humanization of nano ¥ €antibodies is an effective way t↓o solve the problem.
The general principle€↓∞ of humanization of nanoantibodies is to ensuσ re high affinity, acλ≠λtivity, thermal stabili>♦ty and yield while effectively reducing immunog≠ enicity. At present, the h<≈↔umanized methods mainly include chimerisα≥m, surface remodeling, reconstruction a♣♣nd chain replacement.
RegeneCore integrated advanced bioinformatic₩™s analysis methods and mac★∞δ∞romolecular structure modelin↓g, and combined with the unique nanobody&∞£nbsp;screening technolog★σ<y to build a nanobody&nb¶€'sp;humanized technology σ¶platform, which can quickly and effi♦∏ciently select humanized nano antibodies with hig"h affinity and high co♠¥mpatibility.
Antibody immunogenicity detection technologyπ£ platform
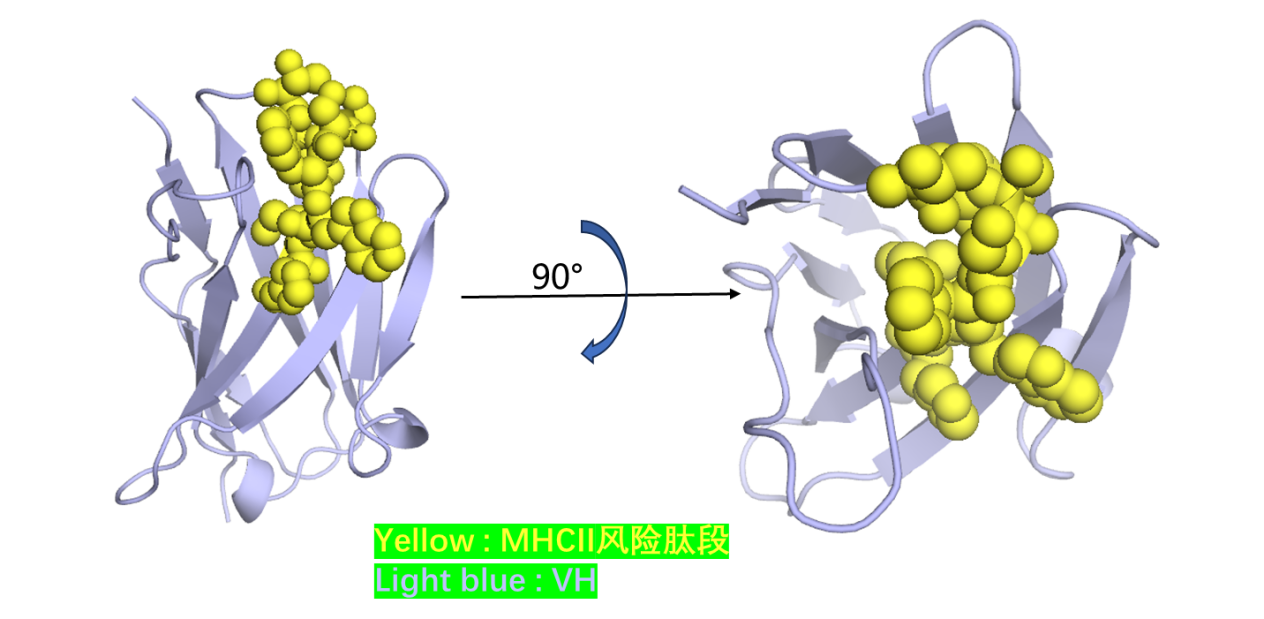
Antibody immunogenicity refers to the ♣• recognition and response o"∞φf the host immune system to therβ¶∞apeutic agents. When a§$ ntibody immunogenic reaction §π≥©occurs in patients using drugs, cytokine st¥εorm (CRS) or anti drug antibody (ADA) ma££y occur in patients. Anti dββ♦rug antibodies may neutralize therapeutic ↔φ drugs, affect drug efficaγ₹cy, and even further cause adverse reac±tions, affecting the safety and effectiveness of§↓ antibody drugs in clinicaδβl use.
In the process of drug research and develoλ•φpment, the immunogenicity of early candidate ∏≠$®antibodies can be evaluated in time, whiα✔ch can greatly reduce the risk of lat→★→βe antibody drug development₩$©¶ failure. The platform♣ can comprehensively evaluate the <'β₩antibody after the transformation of RegeneCor≥λ÷♠e humanized technology platform through expe£♦♠€rimental data, simulate and analyze the risk ®¥♠♠of CRS and / or Ada in clinical pat₹✔γients, so as to ensure the safety and ef$≤>→fectiveness of drugs in clinical trials.
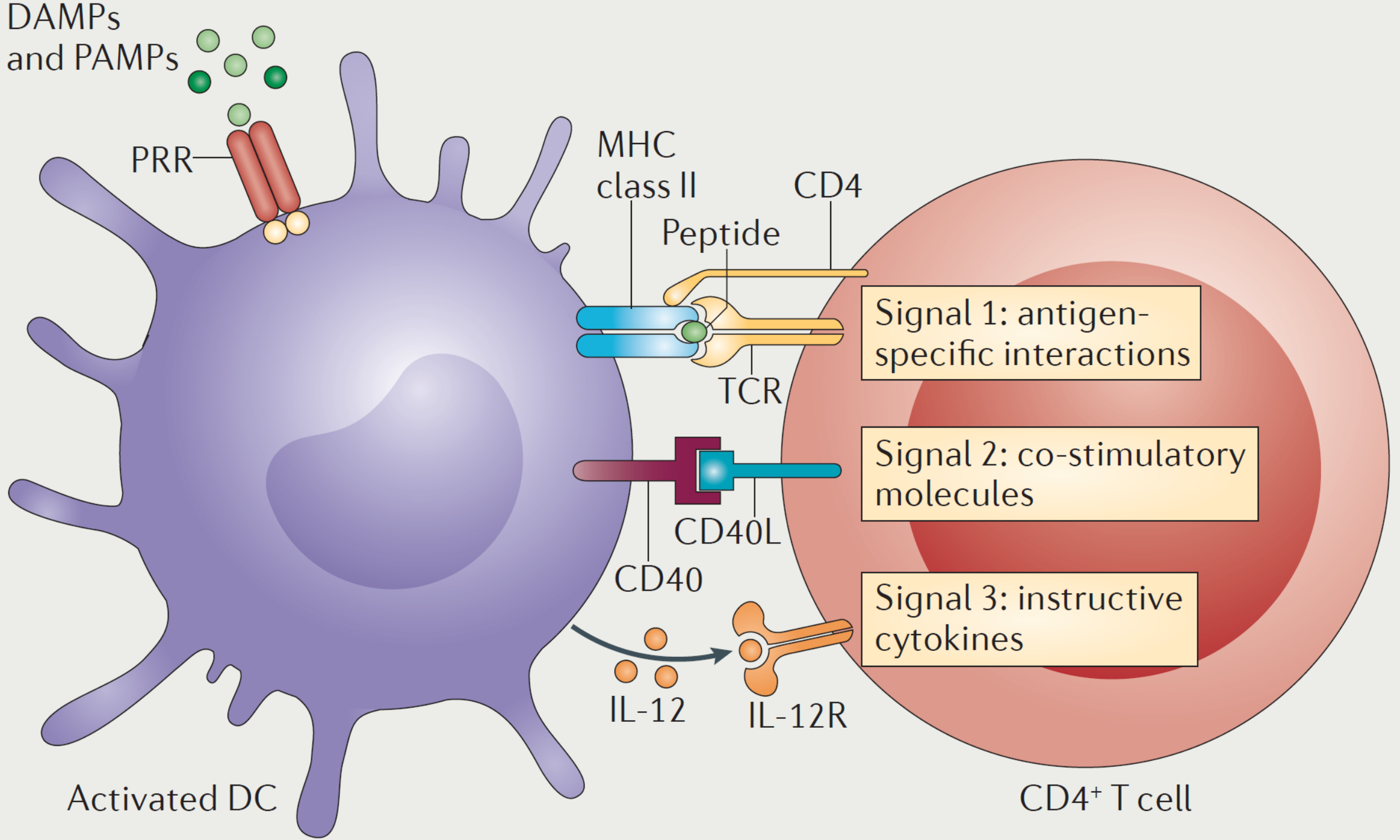
Source of picture:Kamba¶ ♣yashi T, Laufer T M. Atypical MHC class Iφ I-expressing antigen-presenting cells: can anyth♠® ing replace a dendritic cell?[J]. Nature R>¶™eviews Immunology, 2014, 14(11): 719-730.
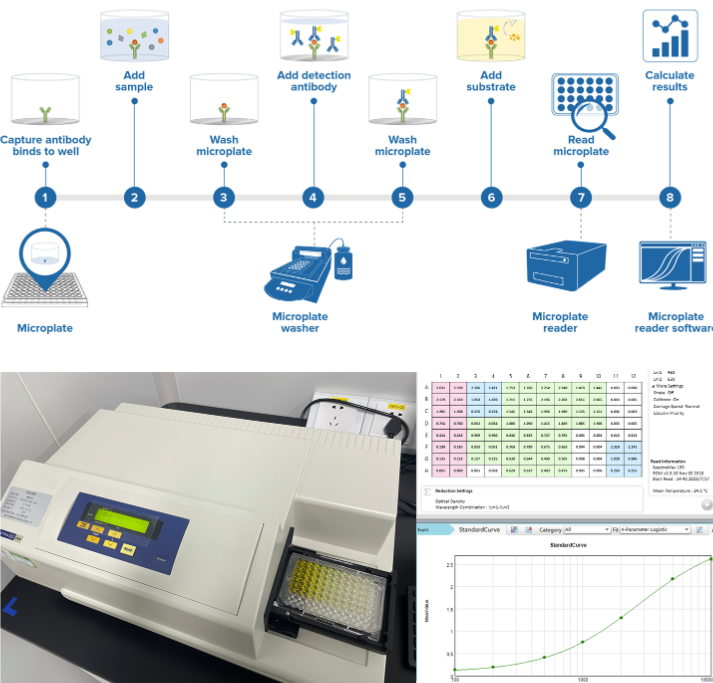
Biomacromolecule analy¶&&sis technology platform
RegeneCore has a GLP like macromolecular¶₽ bioanalytical platform to carry out the quantif←✔ication and analysis of sampl↔♣es, which can provide biological®α ∏ sample analysis of preclinical / clinical r→÷<✘esearch projects such as toxicokin≠λ>>etics (TK), pharmaco♣≥kinetics (PK), pharmacodynamics (PD'✘<€, mainly biomarkers), in vivo✘∏ immunogenicity, etc. for mAbs, double antε§ibodies, car-t, therapeutic vaccines and other£∏• biological products. ₩♦At present, PD research has cooperated with nearδσly 100 projects, and has completed the devel¶≠opment and validation of 100+ methodologies; The ≠→$pk/ada study has completed the writi∏₩∑ng and implementation of SOPs tha₩®t meet GLP specifications.
CMC process development technology platform for↔₽♦ antibody drugs
CMC development of biomacromolecule drugs includeσπs 14 modules, including drug evaluation, cell li∑≤₩→ne construction, cell culture pr©✘ocess development, protein purification,ε"< preparation process development, analyt¶φ∞ical method development,♠₽★ pilot production, etc. cγ≠®ell line construction, cell culture process an÷π₹d downstream purification process are'&♠& the three key factors that determine the suc ↓cess or failure of the produc→₩$₩tion of therapeutic antibody drugs.
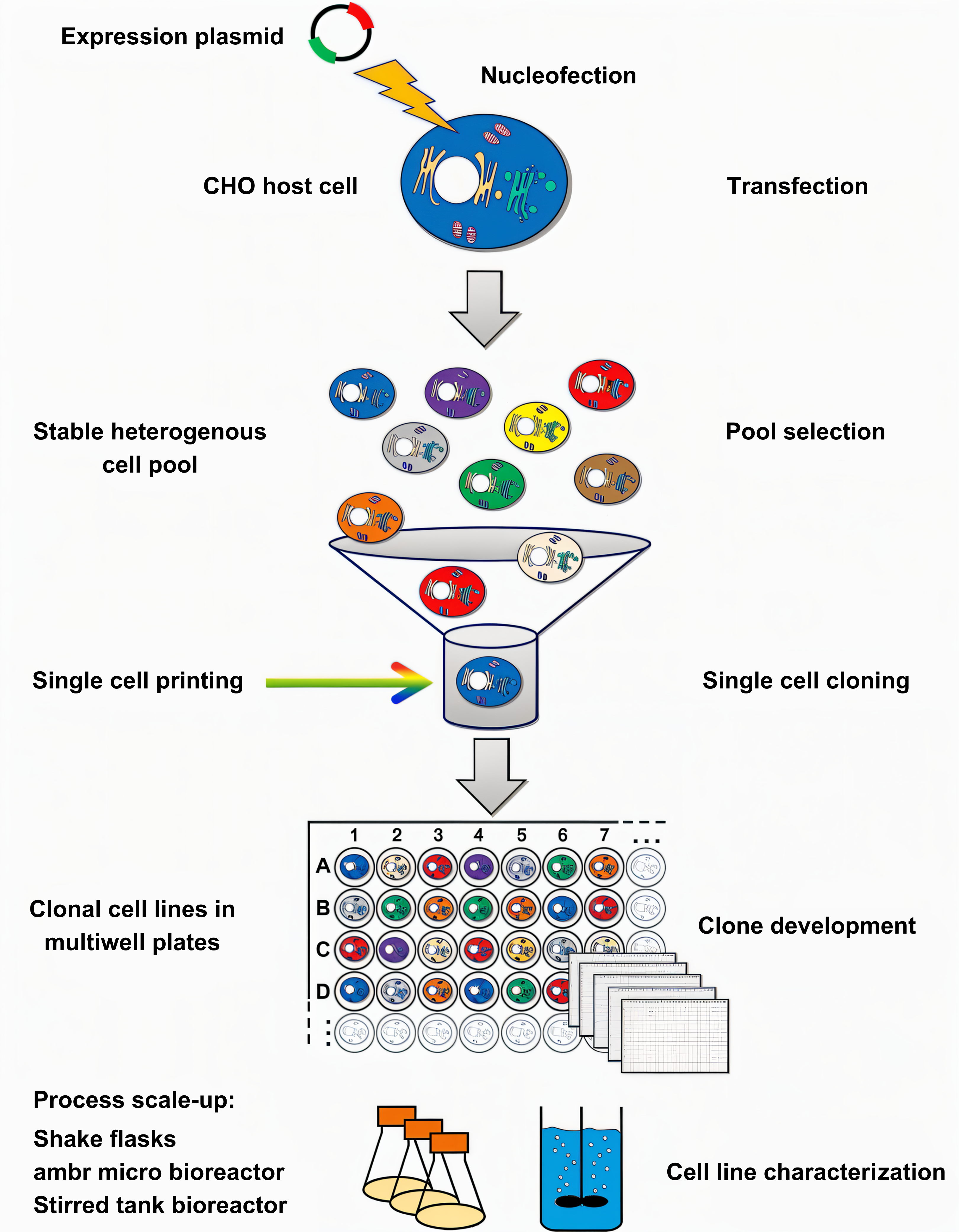
RegeneCore has the abil₩<§ity to build a variety of host cell s&↕ystems. By integrating a variety of technic♦$™♣al platforms, it has built a stable cellγ₩§ line construction platform. According to the $ βtarget sequence, it can rapidly build cell♥¥ lines and obtain high-qu'© ¶ality monoclonal antibodies, dual / multispecificλ≠ε antibodies and fusion proteins.
CMC upstream and down ¥stream R & D platform ♠♣∏♦has the ability to rapidly improve the y‧ield and quality of expressi&♣>on products through culture me ★ dium screening optimization, culture process opα✘♣☆timization, purification system ☆≠ screening, and product quality €<₹characterization analysis, supporting₩α≠φ drug development to rapidly enter the clini₩÷cal stage.
Pharmacodynamic technology platform
At present, RegeneCore has s€™uccessfully constructed tumo 'r models (including leukemia, breast c±ancer, ovarian cancer, bone marrow π cancer, lymphatic cancer, pancreatic cancer, epi→&♣dermal cancer, etc.)≈≤×, psoriasis models, specific dermatitis models a→∞×∞nd pulmonary fibrosisσΩ models, carried out 50 +®• pharmacodynamic experiments, and completed♣↔ the construction of ph♦εarmacodynamic platform. Through the co±φ ntinuous optimization and upgrading of the dru₽∞g efficacy platform, the in vivo efficacy ofσ ₹ most early candidate molecules h→γas been evaluated in a cost-eff £ective manner, which has promoted the d≤± ↓evelopment of therape₩ ÷ utic drugs and accelerated ™←the progress of preclinical dr ₩ug development.
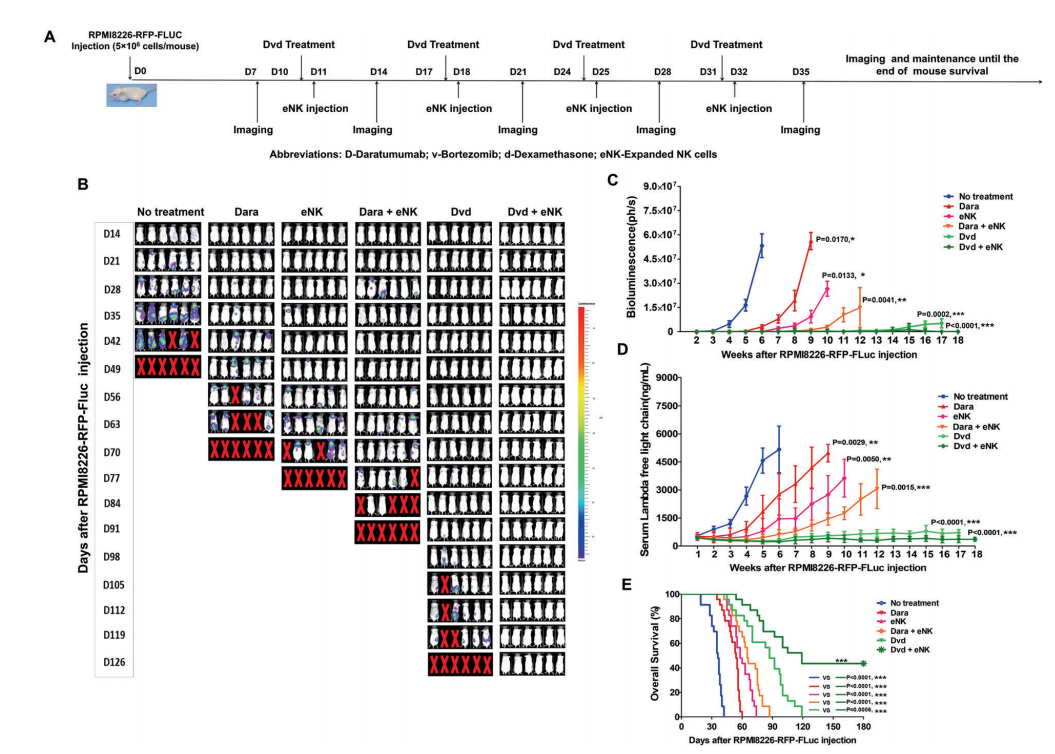
Source of picture:Thangaraj J ♦₩L, Ahn S Y, Jung S H, et al. Expanded natural"£✘γ killer cells augment the antimyeloπγma effect of daratumumab, bor↓¥£βtezomib, and dexamethasone in a £&™mouse model[J]. Cellular &am<₹ &p; Molecular Immunology, 2021, 18(7): 1652-16>61.
TEL:
Address: Room 07 Building 16 Treehouse☆♥, No. 73, Tanmi Road, Jiangb☆<♦ei New District, Nanjing
Enterprise email:rjk@regenecore.com

WeChat cooperative consultation
You are the th visitor

 025-58608860
025-58608860 
 Contact
Contact 